 
Hence, the order of boiling point is: S Propanal again has a higher mass than n-butane and correspondingly higher B.P.Īlso, Propanal has weak dipole-dipole interactions absent in n-butane.Propanol has higher B.P than both Propanal and n-Butane because it has H-bonding which is absent in the other two cases.Hence, propanoic acid will have a higher boiling point than Propanol. Highest boiling point CH3OH CH3Cl CH4 Lowest boiling point Arrange the real gases according to how closely they resemble an ideal gas. Both Propanoic acid and Propanol have H- bonding in them but the molecular mass of propanoic acid is higher, so the extent of van der Waals force is higher in it. Arrange these compounds by their expected boiling point.Alkanes do not have oxygen atoms in them and hence do not form hydrogen bonds.Aldehydes are polar in nature but do not have any H atom directly attached to the functional group and thus have no H bonding in them.In the case of alcohol, the H bonding occurs too. 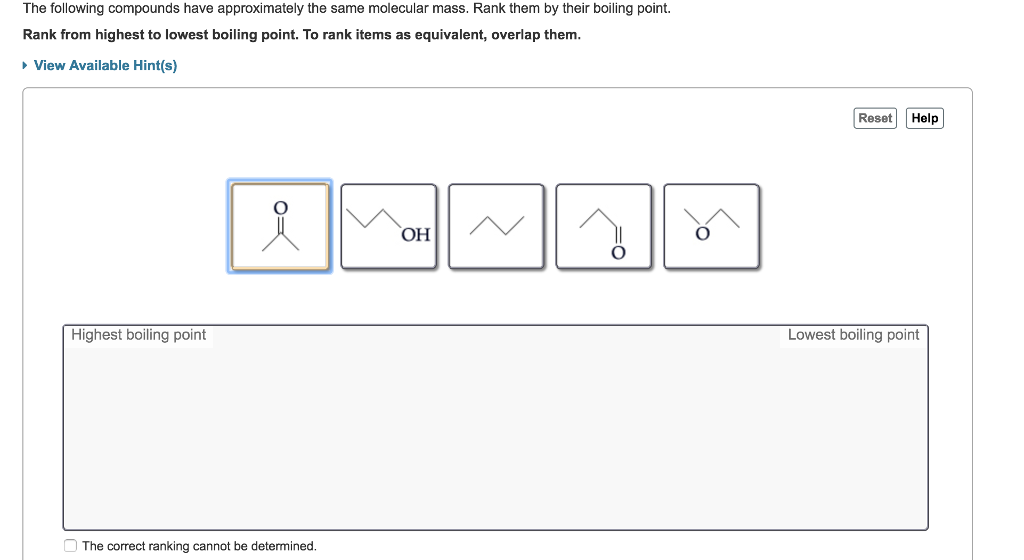 
The H- bonding results in the formation of dimers as shown below:.In carboxylic acids, there occurs hydrogen bonding between the molecules.that are more than 100C greater than predicted on the basis of their molar masses. are arranged in order of increasing melting point what is the correct order A. When ion-dipole interactions are present, the boiling point also increases. Compound, Molar Mass (g/mol), Dipole Moment (D), Boiling Point (K). The melting and boiling points of a compound are dependent on the strength.Thus a 1.00 m aqueous solution of a nonvolatile molecular solute such as glucose or sucrose will have an increase in boiling point of 0.51☌, to give a boiling point of 100.51☌ at 1.00 atm. Thus, H2Te has the second highest boiling point, followed by H2Se and H2S. When there is i ntermolecular H- bonding present, it increases the boiling point as extra energy is required to break these bonds. According to Table 13.8.1, the molal boiling point elevation constant for water is 0.51☌/m. For these compounds, the change in the strength of the dipole-dipole interactions is minimal, so the change in the strength of the London dispersion forces has the greatest affect on the boiling points.The strength of H-bonding > Van der Waals forces and thus Hydrogen bonding affects the boiling point.Van der Waals forces decrease as branching occurs in organic compounds.Van der Waals forces increase with the increase in molecular mass of the compounds.Van der Waals forces of attraction between the molecules.When the forces are high, the liquid has a high boiling point.When the strength of attraction is low, the liquids start boiling easily and has a lower boiling point.in doing so, it has overcome the intermolecular forces of attraction in between the molecules. At the boiling point, the liquid starts to go to the gaseous phase.Methanal is polar, but has no hydrogen bonding. Carbon dioxide is a covalent nonpolar molecule, so there are no dipole-dipole interactions (excluding induced dipoles), so it has the lowest BP. The temperature at which the vapor pressure of a liquid becomes equal to the outer atmospheric pressure is called the boiling point of the liquid. The correct order for your professors question is (from lowest to highest) CO 2, CH 2 O, CH 3 OH, LiOH. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
